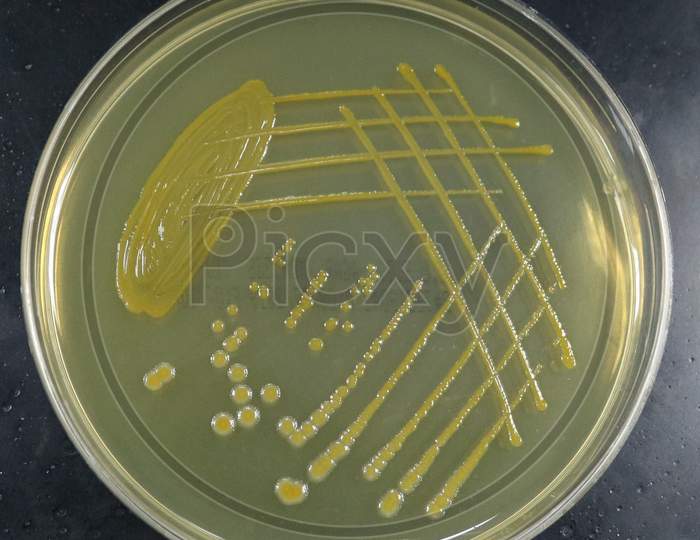
The use of in-line suction to collect a specimen.Tracheal Aspirate Cultures: The Challenges So, what is so challenging about these cultures? The truth is, the inconsistencies begin with specimen collection and continue all the way through to susceptibility reporting. However, guidelines have never been established, and as a result, these cultures remain unreliable, variable and costly. Two studies performed in the 1990’s proposed Gram stain rejection criteria for tracheal aspirate specimens with >10 squamous epithelial cells or no organisms detected on the Gram stain for adults and the absence of organisms on Gram stain for the pediatric population. Laboratories must provide evidence that they are using some form of Gram stain rejection criteria and not setting up culture on specimens deemed inappropriate by these criteria.Ĭurrently, there are no standards for laboratory workup of tracheal aspirate cultures.This must be documented in a laboratory procedure. Gram stains should be routinely performed to determine specimen acceptability and used as a guide for culture workup.To support the use of these criteria, the College of American Pathologists (CAP) requires the following when inspecting a clinical laboratory for compliance: 
In general, the criteria suggest that a specimen with a high number of epithelial cells by Gram stain is contaminted and is likely unacceptable for culture. Rejection criteria for sputum samples is still broad and not universal, but they have provided algorithms for laboratories to follow in order to prevent the culture of samples that are grossly contaminated with oropharyngeal flora. Reducing the number of ventilator-days per patient could make ventilators available for the next patient in need sooner, and interpreting tracheal aspirate culture results with caution could prevent unnecessary side-effects from antimicrobial use.Ī Brief History of Respiratory Specimen Rejection Criteria While respiratory samples will always be a challenge, not all respiratory specimens are without guidance. It is estimated that the United States only has 62,000 modern ventilators, far less than the anticipated number of severe COVID-19 cases that are expected to occur. It is especially important to manage mechanically ventilated patients as appropriately as possible in the time of SARS-CoV-2/COVID-19. Even though the tracheal aspirate culture method lacks accuracy in diagnosing VAP, clinicians are left with little choice but to use it given that other diagnostic methods are limited and other specimen types are more difficult to obtain. This overuse has been correlated with the emergence and spread of antibiotic-resistant organisms in the ICUs of both adult and pediatric institutions. Ultimately, presumed VAP results in high resource utilization and is the most common reason for empiric antibiotic use in the ICU. Diagnostic uncertainty leads providers to empirically treat for VAP, regardless of whether the patient is confirmed to have it or not. Of the 3, tracheal aspirate specimens are easier (and potentially safer) to collect, but they have low diagnostic specificity for VAP and rarely distinguish between colonizing microbiota and microbiota causing infection. The serious consequences of this disease demand quick and accurate diagnostic testing, yet no gold standard exists.Ĭriteria provided by the Centers for Disease Control (CDC) recommend 3 diagnostic methods for VAP: bronchoalveolar lavage (BAL), lung biopsy and tracheal aspirate. 
VAP is the second-most common hospital-acquired infection in intensive care units (ICUs), and the most common cause of nosocomial infection leading to death in critically ill patients. Mechanical ventilation is a life-saving measure used on thousands of patients in the United States each year, but patients that receive this intervention are at an increased risk of developing a severe condition called ventilator-associated pneumonia (VAP).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
